-
Strive to Improve Medical Conditions, Reduce Health Care Costs.
9 Renmin Rd(E), Yangshe Town, Zhangjiagang City, Jiangsu Province, China.
+86 18913609266
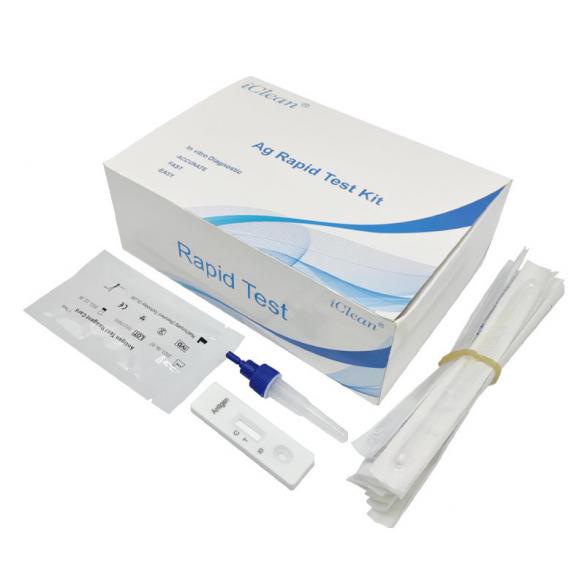
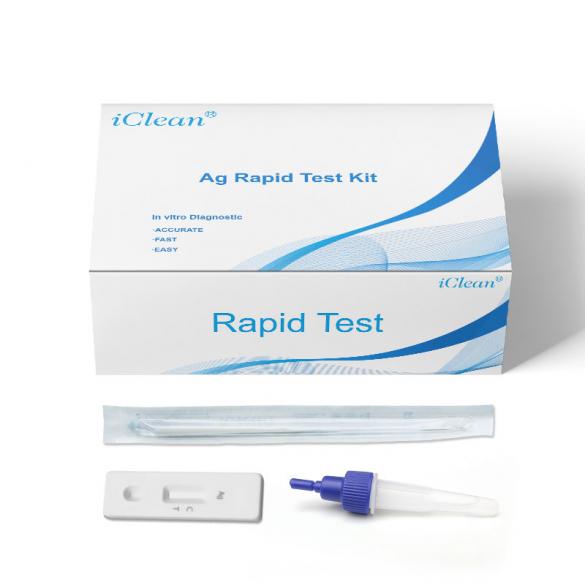
COVID-19 Antigen Rapid Test Kit (25-Pack): Oral/Nasopharyngeal Swab Test
During the detection process, the gold-labeled anti-coronavirus monoclonal antibody on the labeling plate binds to the neocoronavirus antigen in the sample to form a complex, and the reaction complex moves forward along the nitrocellulose membrane under the action of chromatography, passing through the nitrocellulose.
The detection area (T) on the plain film is pre-coated with an anti-2019-nCoV monoclonal antibody, and finally a red reaction line is formed in the T area. If the sample does not contain the 2019-nCoV antigen, a red reaction line cannot be formed in the T zone. Regardless of whether the test sample contains the 2019-nCoV antigen, a red reaction line will always be formed in the quality control area (C).
Product Description
The COVID-19 Antigen Rapid Test (Lateral Chromatography) is an in vitro immunochromatographic method for the qualitative detection of SARS-CoV-2 nucleocapsid protein antigens from flocked nasopharyngeal (NP) or nasal (NS) swabs of individuals suspected of COVID-19. This reagent uses the double antibody sandwich method to legally detect the novel coronavirus antigen in nasopharyngeal and oropharyngeal swabs. It is designed to assist in the rapid diagnosis of SARS-COV-2 infection.
This test is limited to laboratories that are certified to perform medium, high complexity, or exempt complexity testing requirements. The test is authorized for use at Point of Care (POC), which are inpatient care facilities that have received CLIA exemption certificate, compliance certificate, or certification certificate. The test is to identify SARS-CoV-2 nucleocapsid antigen that is generally detectable in upper respiratory samples during the acute phase of infection. A positive test result indicates the presence of viral antigens, but the correlation of clinical, medical history and other diagnostic information is necessary to determine infection status. However, bacterial infection or co-infection with other viruses can also cause a positive test result.
Since it cannot be ruled out that the subject is only infected with SARS-CoV-2, a negative test result should be regarded as a presumed result. It cannot be used as the only basis for treatment, patient management, and infection control. Negative test results should be considered based on the patient’s recent exposure history, medical history, and clinical signs and symptoms consistent with COVID-19. They should be confirmed by molecular testing for patient management if necessary.
COVID-19 Antigen Test Kit(Lateral Chromatography) is intended for use by trained clinical laboratory personnel specifically instructed and trained in the techniques of in vitro diagnostic procedures and proper infection control procedures and individuals similarly trained in point of care settings.
Product Specification: 25pcs/box
How To Use?
Application: For suspicious patients with symptoms, mild symptoms, or even without symptoms, also for testing people with close contact with infected patients and people under quarantine control.
The assay is for the direct and qualitative detection of antigen of SARS-CoV-2 from nasopharyngeal secretions and oropharyngeal secretions specimens.
Delivers clinical results between 15 and 30 minutes
Visual interpretation of results
No special equipment is needed
Product Instructions
Sample Collection and Preparation
Set up for the test
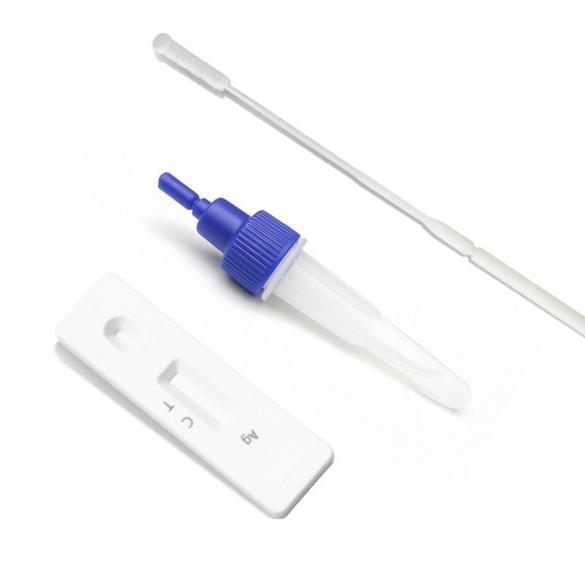
Remove one extraction tube and one COVID-19 Antigen Test Kit(Lateral Chromatography) cassette from its foil pouch immediately before testing.
Label the test kit and the extraction tube for each specimen to be tested. Place the labeled extraction tube(s) in a rack in the designated area of the workspace.
Nasal Swab Specimen Collection
When taking a nasal swab sample, carefully insert the swab (attached in the kit) into the nostril where the most secretions are observed with the naked eye. Gently rotate and push the swab to the point of resistance at the turbinate bone (approx. 2.0-2.5cm or 1 inch near the nostril), then roll the swab on the nasal wall approximately five times before removal.
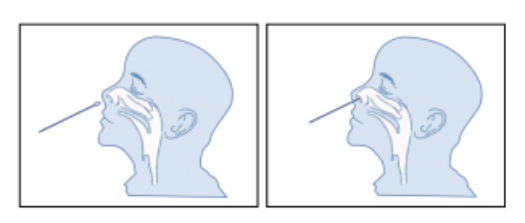
Sample Preparation
After the specimen collection is done, immediately immerse the swab into the sample extraction buffer.
To make the sample extracting completely, permeated into the buffer from the swab, rotate the swab against the tube wall repeatedly for 10 seconds and then use fingers to hold the tube (the tube wall is semi-solid) to squeeze the swab several times while slowly removing the swab out of the tube.
The purpose of swab squeezing against the tube wall is important because it can keep the specimen containing liquid to remain in the tube as much as possible. After taking out the swab and discarding it into a biohazard waste container, put on a nozzle (supplied in the kit) to the top of the extraction tube tightly and gently shake the tube to mix the inside liquid well.
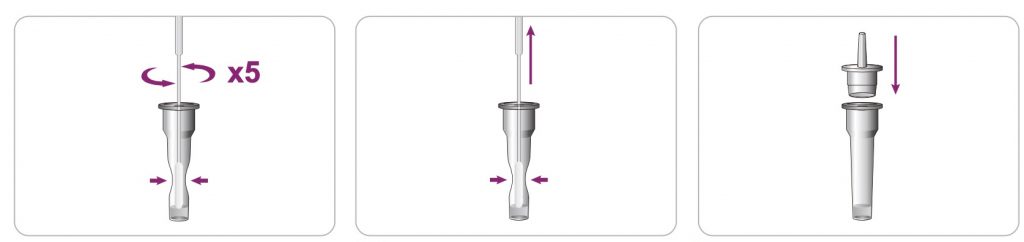
Test Procedure
1. Read the instruction for use carefully before performing the test.
2. Reverse the specimen extraction tube and keep it upright. Squeeze the test tube, and add more than 3 drops of the specimen liquid (about 70-90 μL) into the test cassette through the test tube nozzle.
3. Start the timer. Read the result visually at 15 minutes. Do not interpret the result after 30 minutes.
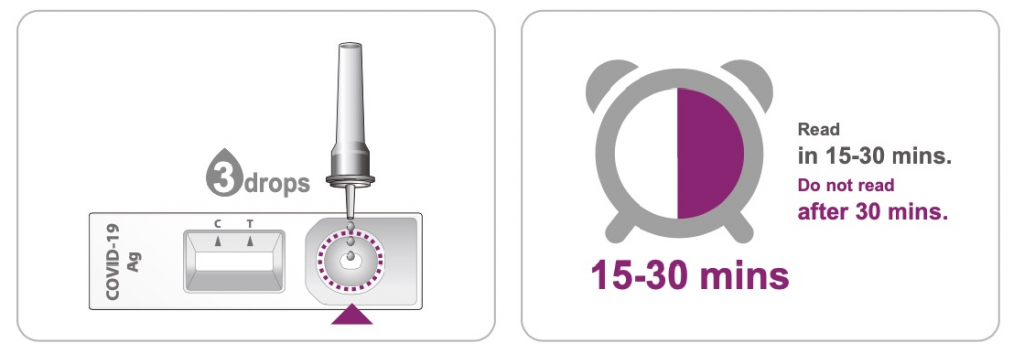
Interpretation of the Results
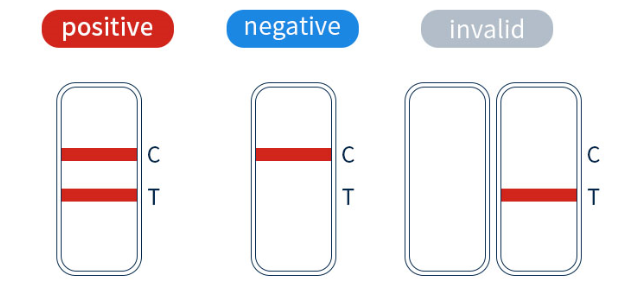
Positive test results: Visible lines of color appear at both the “T” and “C” sites, or if the test “T” site shows a darker color.
Negative test results: Only the “C” reference has color, and the “T” position is blank.
Invalid result: If “C” is empty and no color appears, the test result is invalid, so the sample needs to be tested again.
Performance Characteristics
Sensitivity: 96.15%(100/104), confirmed positive cases compared to the nucleic acid amplification test.
Specificity: 99.78%(448/449), the negative cases were also confirmed by nucleic acid amplification test.
Cross-reactivity: There is no cross-reactivity with influenza A virus, influenza B virus, adenovirus, Coxsackie virus, ECHO virus, and enterovirus; no cross-reactivity with Chlamydia pneumoniae, Mycoplasma pneumoniae, Chlamydia psittaci, and Chlamydia trachomatis; no cross-reactivity with Acinetobacter baumannii, Bordetella pertussis, Candida albicans, Escherichia coli, Haemophilus influenzae and Neisseria gonorrhea.
Limitations of Procedure
1. The test kit is used to directly detect the presence of SARS-CoV-2 antigen in respiratory specimens from patients suspected of covid-19.
2. This test kit is a qualitative assay, not a quantitative assay.
3. The accuracy of the test depends on the sample collection process. Improper sample collection, improper sample storage will affect the test result.
4. The results of this test kit are only intended for clinical reference. A confirmed diagnosis should only be made after all clinical, and laboratory findings have been evaluated.
5. Due to the limitations of antigen detection test kits, it is recommended to use nucleic acid amplification or virus culture identification methods to review and confirm negative test results.
6. Positive test results do not rule out co-infections with other pathogens. A negative result of this test kit can be caused by:
1) Improper sample collection, improper sample transfer, or handing.
2) The level of the SARS-CoV-2 virus is below the detection limit of the test.
3) Variations in viral genes that may have caused changes in antigen determinants.
CAUTIONS
This kit is only used for in vitro auxiliary diagnosis and should be used strictly by the Instructions For Use.
Please check the kit’s effective date and package integrity before use. The test equipment cannot be used if its packaging is damaged or the product is outdated.
The time for testing and results reading must be followed.
After the pouch of the test cassette is opened, the test should be performed within 60 min. The test cassette can only be used once.
The kit is stored at 20℃~30℃. Keep away from moist, sunlight, heat, or freezing conditions.
The test results of this kit are only for clinical reference, and the clinical diagnosis of the disease should be considered in combination with the patient’s symptoms, signs, medical history, other laboratory tests, and treatment response.


Leave a Reply